
Also check out the News and Views by Ayse Ecer and Tatjana Trcek!
nature.com/articles/s41...

Also check out the News and Views by Ayse Ecer and Tatjana Trcek!
nature.com/articles/s41...

New preprint from the Trcek lab, with Siran Tian as the lead author - congratulations Siran! doi.org/10.64898/202...
4) nanos mRNA localization counteracts Vasa-mediated enhancement of Oskar mobility. While germ granule mRNA localization requires Vasa, this process opposes Vasa’s effect on Oskar condensates, providing an RNA-mediated negative feedback mechanism that regulates condensate properties.
3) mRNA localization to Oskar condensates requires Vasa and its interaction with Oskar. Although germ granules contain diverse proteins, Vasa and Oskar are necessary and sufficient for robust germ granule mRNA localization to the granules.
2) Vasa’s modulation of condensate properties requires direct protein-protein interaction. This activity requires specific binding interfaces between Vasa and Oskar, but is less dependent on Vasa helicase activity.
1) Vasa modulates the material properties of ribonucleoprotein condensates in fly oocytes and human cells. Recruitment of Vasa increases the mobility of the germ-granule nucleator Oskar, thereby preventing aggregation of granules.
This new function in regulating protein condensation extends beyond its established function as an RNA helicase. Specifically, Siran shows that:
She investigated the role of a conserved DEAD-box RNA helicase Vasa in condensation of germ granules in Drosophila. Her work reveals a previously unrecognized function of Vasa in regulating the material properties of Oskar ribonucleoprotein condensates through its direct interaction with Oskar.

New preprint from the Trcek lab, with Siran Tian as the lead author - congratulations Siran! doi.org/10.64898/202...
4) Germ granule mRNAs are replete with GC-rich complementary sequences predicted to be competent of engaging in persistent intermolecular base pairing however in vivo these interactions are unrealized and restricted by RNA folding.
3) Engineered germ granule mRNAs with exposed GC-rich complementary sequences (CSs) within stem loops induce sustained base pairing in vitro and enhanced intermolecular interactions in vivo. But, the presence of these structures disrupts development, and GC-rich CSs exacerbate this phenotype.
We find that:
1) mRNAs remain structured within germ granules
2) mRNAs base pair intermolecularly without high sequence complementarity or significant melting of secondary structure. Base pairing is driven by scattered and discontinuous stretches of bases appearing on the surface of structured RNAs

New paper from the Trcek lab, with Siran Tian as the lead author - congratulations Siran!: www.nature.com/articles/s41...

New preprint from the Trcek lab - Jeffrey Liao as the lead author! We studied intermolecular base pairing using AMT crosslinking in mammalian cells during oxidative stress doi.org/10.1101/2025...
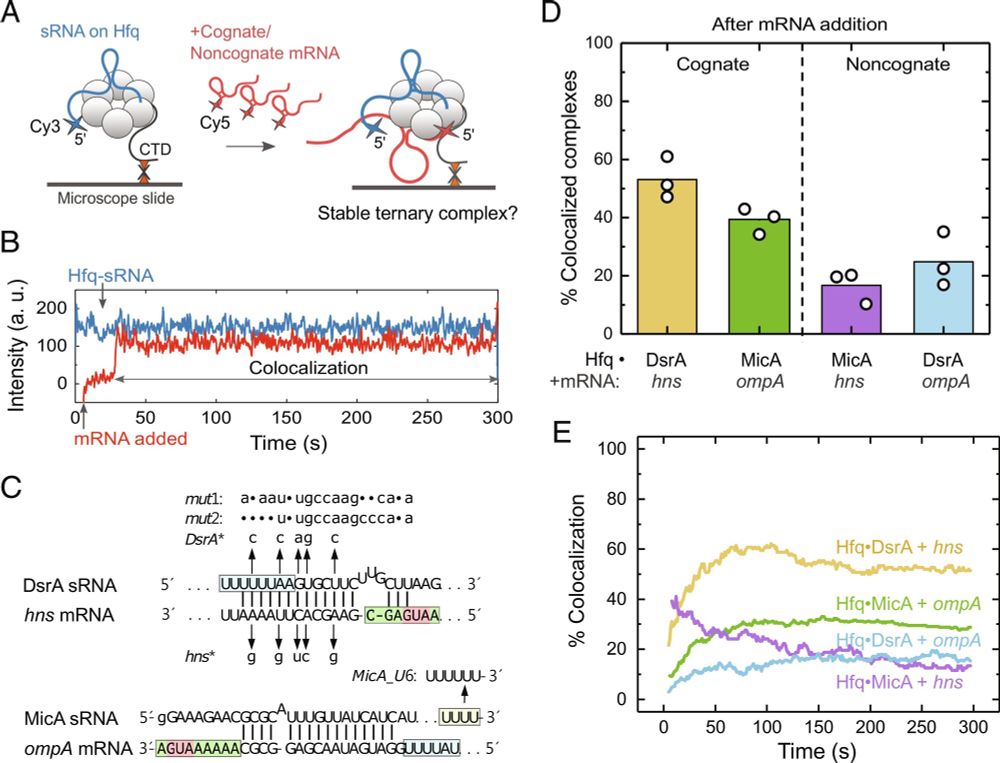
Our new paper is out in PNAS! We show that competition for binding to the Hfq matchmaker disfavors mismatched sRNA-mRNA combinations while allowing matched sRNA-mRNAs to form stable pairs. Kudos to Jorjethe Roca and Yi-Lan Chen for their fantastic work on this problem! doi.org/10.1073/pnas...
Congrats, Jorjethe and Yi-Lan! 👏👏

New method drop:
HCR-Proxy, a modular proximity labelling approach to profile local proteome composition around RNA at nanoscale, subcompartmental resolution. Thereby we resolved nested nucleolar layers and uncovered the grammar of spatial protein partitioning.
www.biorxiv.org/content/10.1...

Excited to share our lab's preprint! We found that flexible RNAs have much broadened ion atmospheres than structured RNAs. This leads us to speculate the implications on molecular recognition and phase behavior. Please check full text for other interesting details www.biorxiv.org/content/10.1...
Jeffery is presenting his work at the annual RNA society meeting in San Diego - visit his poster! #RNA25; #RNA2025
Jeffery is presenting his work at the annual RNA society meeting in San Diego - visit his poster! #RNA25; #RNA2025
Characterization of intermolecular base pairing using AMT crosslinking in mammalian cells during oxidative stress https://www.biorxiv.org/content/10.1101/2025.05.21.655305v1
Characterization of intermolecular base pairing using AMT crosslinking in mammalian cells during oxidative stress https://www.biorxiv.org/content/10.1101/2025.05.21.655305v1
We propose that the combined suppression of intermolecular base pairing and the increased RNA structural diversity may serve as a mechanism to preserve normal RNA function during stress, while also facilitating the reversible assembly of stress granules
c) However, these RNAs increase in structural diversity in a manner dependent on oxidative stress and the stress granule nucleating proteins G3BP1 and G3BP2.
d) In the absence of the main stress granule nucleating proteins G3BP1 and G3BP2, mRNAs remain dispersed in cells during oxidative stress
We show that:
a) Oxidative stress downregulates intermolecular base pairing of reporter mRNAs engineered to enhance detection of such interactions.
b) RNA regions within transcripts that enrich in stress granules remain structured during stress.

New preprint from the Trcek lab - Jeffrey Liao as the lead author! We studied intermolecular base pairing using AMT crosslinking in mammalian cells during oxidative stress doi.org/10.1101/2025...