Our organocatalytic stereoselective head-to-tail macrocyclization is now out in @science.org! We use a peptide catalyst to access 12- to 18-membered rings featuring various functionalities with catalyst-controlled stereochemical outcome. #chemsky
Our organocatalytic stereoselective head-to-tail macrocyclization is now out in @science.org! We use a peptide catalyst to access 12- to 18-membered rings featuring various functionalities with catalyst-controlled stereochemical outcome. #chemsky

Great start to the week with our latest carpyridine findings out in @jacs.acspublications.org! Water and carpys now self-assemble together, forming linear stacks as revealed by micro-ED. Check it out: doi.org/10.1021/jacs...
@michelrickhaus.bsky.social @avj-chem.bsky.social @rickhauslab.bsky.social

🚨 New preprint from our group! We report a photocatalytic, regiodivergent method to functionalize saturated N-heterocycles at the α- or β-position. The metal-free reaction runs in water with a flavin catalyst and gives rapid access to sought-after piperidines. Check it out here: 🔗 tin.al/DqxU
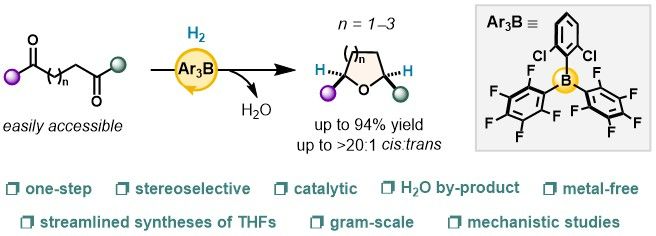
🚨 Happy to share a new preprint – our group’s first!
🔗 chemrxiv.org/engage/chemr...
We report a novel synthetic method to access cyclic ethers from diketones, powered by simple borane catalysts.
Thanks to the whole team for their excellent work!