Super happy to receive an M-2 weave grant from the NWO (Dutch Research Council). Together with Sayed Abd El-Whab we will study the molecular features of H5Nx HA and NA proteins. #glycotime www.nwo.nl/en/news/gran...
Super happy to receive an M-2 weave grant from the NWO (Dutch Research Council). Together with Sayed Abd El-Whab we will study the molecular features of H5Nx HA and NA proteins. #glycotime www.nwo.nl/en/news/gran...

#glycotime alert! Zhiyong synthesized O-acetylated GD3 gangliosides, which were used to examine viral receptor specificities in a cellular context by @LiangRuonan great work by @zeshi_li and @TheBoonsGroup @uu-cbdd.bsky.social www.biorxiv.org/content/10.1...
Big shout out to @LiangRuonan and many thanks to @TheBoonsGroup , @UnioneLuca @UU_Glycoscience @uuuips.bsky.social
The latter is vital, as many H3N2 viruses bind solely to these complex structures, and many zoonotic avian HAs, engineered to bind human-type receptors, do so as well.
Significant observations indicate that the conserved 195Y, which changes to F, rescues binding properties in specific backgrounds. And symmetric N-glycans with tri-LacNAc arms are not displayed on human tracheal epithelial cells.
She followed those results up with, amongst others, a tissue binding experiment on human tracheal tissues. As glycan binding is one thing, but binding tissues is another.
Here, we aimed to identify the molecular determinant of tri- vs. di-LacNAc binding in strains that have circulated over the last 10 years. Ruonan created a mutant library and utilized complex N-glycans in an ELISA assay to assess specificity and avidity.
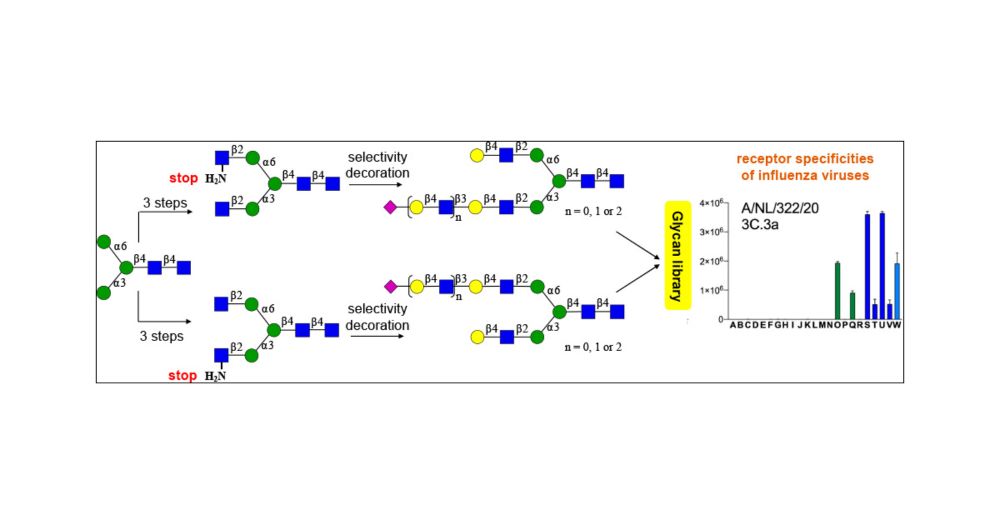
We previously demonstrated that human H3N2 viruses can have very strict specificities to complex N-glycans and that this can very depending on antigenic pressure pubs.acs.org/doi/full/10.... www.nature.com/articles/s41...
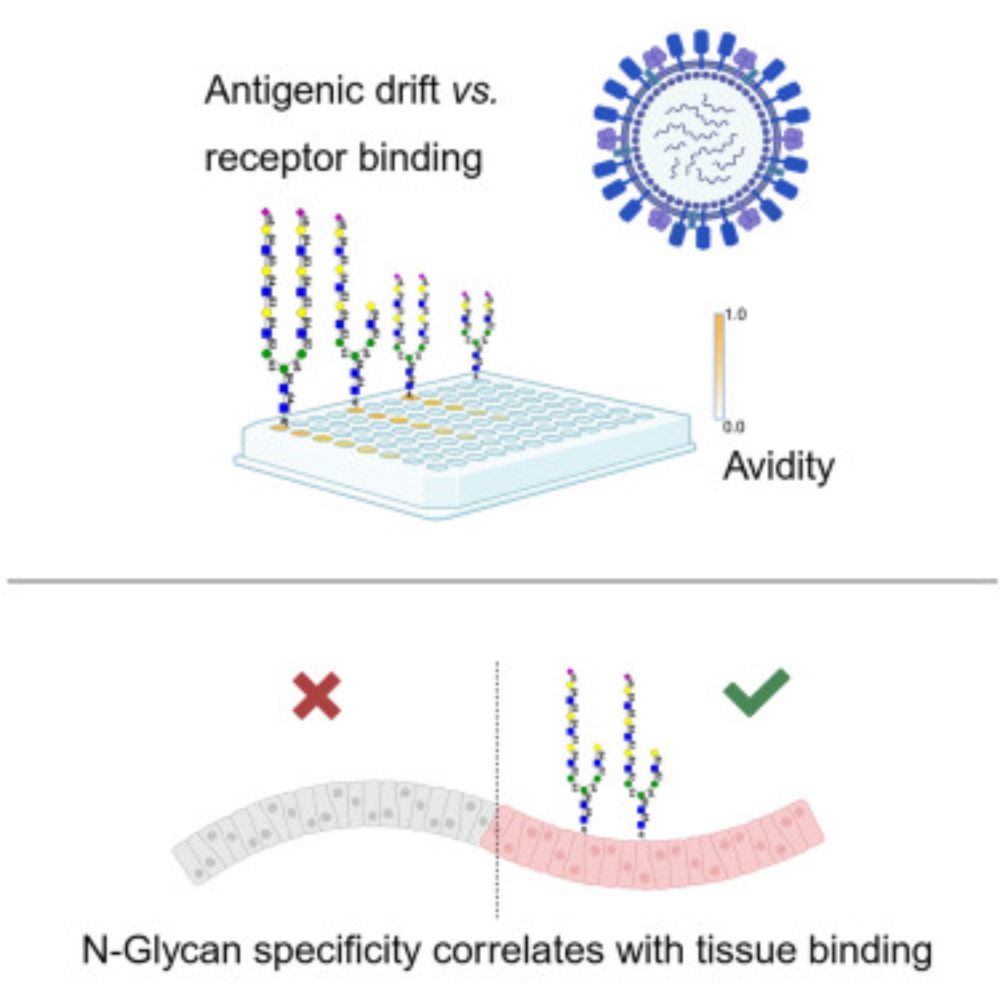
Very happy that our paper on the receptor binding properties of contemporary H3N2 viruses is now published @CellReports # glycotime www.cell.com/cell-reports...
And now published! #glycotime "Sialic Acid-Containing Glycolipids Extend the Receptor Repertoire of Enterovirus-D68" fantastic collaboration with @lisabauervirus.bsky.social @debbyvanriel.bsky.social @TheBoonsGroup enjoy 👍https://pubs.acs.org/doi/10.1021/acsinfecdis.5c00063

We are happy to share the first publication of our consortium:
pubs.acs.org/doi/10.1021/...
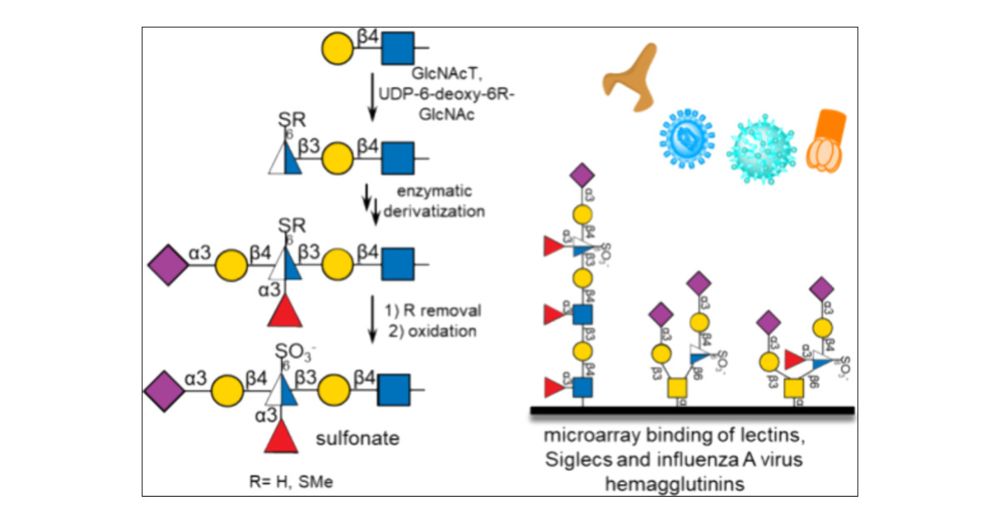
New paper of Yunfei! Here, he synthesized sulfonates in different sialylated epitopes... although not better ligands for most lectins tested, they were tolerated and even preferred by siglec-4. #glycotime pubs.acs.org/doi/10.1021/...
What a great thread and very interesting preprint 👍

Happy Eurovision! Basel is awesome, getting ready for the final!🏳️🌈🇫🇮
Bijvoet symposia 👍 #glycotime @uu-cbdd.bsky.social

Happy to be able to contribute to this very cool preprint
Characterization of the glycoproteins of novel fish influenza B-like viruses #glycotime #lovevirology @uu-cbdd.bsky.social www.biorxiv.org/content/10.1...
Many thanks to @uu-cbdd.bsky.social @uuuips.bsky.social
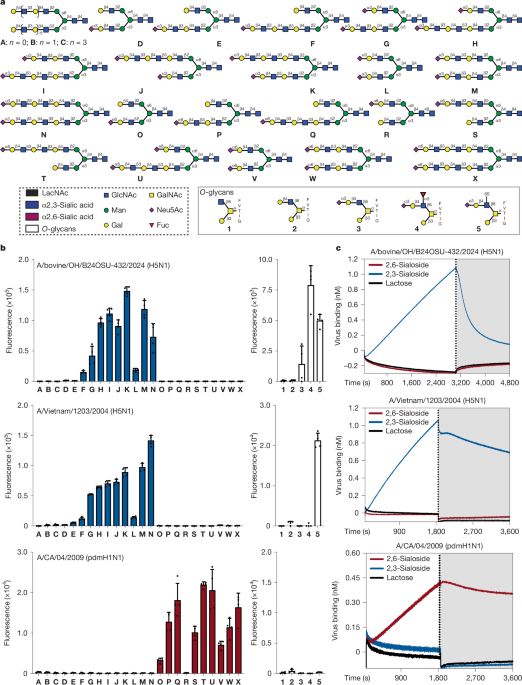
Of course, we don't know if precisely these O-glycans enable these viruses' unique zoonotic capabilities, as many other virological and ecological factors are likely involved. However, we do like to hypothesize that these binding properties contribute to these capabilities.
Another observation we made is that the cattle-derived recombinant virus had nearly identical receptor binding properties compared to one of the first 2.3.4.4 viruses isolated over 10 years ago. Receptor specificity is relatively conserved for 2.3.4.4 viruses.
Our colleagues at @scripps.edu determined the structural features of these specificities. And i hope you can appreciate how the fucose lies in the receptor binding site. Fun observation is that we saw the peptide interacting with a very well-known epitope in the HA stalk
Using these mucin-like O-glycans as a glycan array screen, we observed that these 2.3.4.4b were unique in their promiscuous receptor binding properties. These properties resulted in binding to human respiratory tract tissue while not binding human-type receptors.
Many years ago, a carbohydrate chemist, Julia, started to synthesize mucin-like O-glycans, as they were greatly understudied. This was also when 2.3.4.4b H5Nx viruses were taking the stage.

Excited to have this story out on bioRxv! #glycotime #lovevirology #H5N1 The receptor binding properties of H5Nx influenza A viruses have evolved to promiscuously bind to avian-type mucin-like O-glycans.
www.biorxiv.org/content/10.1...
Now published in PNAS, this is a fantastic paper on how HA moves within a virion upon receptor binding. By the way, do check the #badglycans in Figure 6 😐
www.pnas.org/doi/10.1073/...

We a looking for a Postdoc!
Interested in virology, stem-cell derived models, glycans?
Apply before 29th of October!
@thijskuiken.bsky.social @lisabauervirus.bsky.social @marionkoopmans.bsky.social @rpdevrieslab.bsky.social
www.werkenbijerasmusmc.nl/en/vacancy/1...
Now out in Nature! Bovine H5 maintains binding to avian-type receptors. #glycotyme #H5N1 @uu-cbdd.bsky.social
www.nature.com/articles/s41...

Many thanks to
@TheBoonsGroup @UU_Glycoscience @UUBeta, and if you do not have access, please check the preprint www.biorxiv.org/content/10.1...
Maria and our colleagues at @scrippsresearch have done fantastic work on this. A 2.3.4.4e H5 only needed a single Q226L mutant to bind canonical human-type receptors! A representative H5 from the current cow outbreak did not shift receptor binding with the Q226L only.
Happy to have this story now published in @PNAS
The Q226L mutation can convert a highly pathogenic H5 2.3.4.4e virus to bind human-type receptors | PNAS #glycotime www.pnas.org/doi/10.1073/...
#Viruseslikeitsweet is online! We are a consortium of the department of Viroscience @erasmusmc.bsky.social and the department of Chemical Biology & Drug discovery @utrechtuniversity.bsky.social
Follow our progress here or via our Linked-In account: NWO-XL Consortium Viruses like it Sweet